In the Part 1 of the Soap Story I was talking about the importantce of solid soap and how does it work to remove dirt from our hands and clothes. Even if Soap exist for quite a very long time, it is more a commercial product of the 20th century. Today we use soap on daily basis constanlty. We obviously care a lot about clean clothes. But we also want clean, shiny and fresh-smelling hair. Enter shampoos.
In this Part 2 of the Soap Story I will to talk about liquid soap and shampoos.
The word shampoo came to the English language from India, where it referred to a type of head massage using oil and lotions. The practice was imported to Britain during colonial times and eventually came to mean a type of hair washing. The first modern shampoo was created in the 1930s by Procter & Gamble; it was called Drene.
Drene was made with new, milder liquid surfactants, and was packaged in a glass bottle with a bright green-and-purple label. About the same time Unilever, Procter & Gamble’s primary rival, came into business. Rivalry between these two global firms has led detergent innovation ever since. If you look at the ingredients in a bottle of modern shampoo, you are likely to see something called sodium lauryl-surphate, or its cousin sodium laureth sulphate. These are the building blocks of most modern shampoos; they’re both very effective surfactants that do not interact strongly with calcium water and so do not form scum. They also do something else that we’ve come to consider an essential part of shampoo – they foam. And they do it very, very well.

When you’re using shampoo, foam is created while you’re scrubbing trapping air in water as you lather. The air tries to escape from water, and when it reaches the surface of the liquid, it forms a bubble. If you’re scrubbing your hair without any surfactants, the bubble will just be a thin film of pure water, which has a high surface energy with air, and so it will quickly pop. But all that changes when you add a surfactant like sodium lauryl sulphate to the mix. The surfactant’s molecules will easily collect in the thin film of water that surrounds the bubble, lowering the surface energy of the liquid so much that the liquid film becomes relatively stable. As you massage the shampoo into your hair, these more resilient bubbles will keep forming, resulting in a build-up of foam. Because the surfactant is simultaneously gathering up all the oil and grease, we associate the cleaning with the foaming, and judge the effectiveness of a shampoo by its foaminess. Modern adverts stress this, but the foam does not help the shampoo to clean more thoroughly. Its role is purely aesthetic.
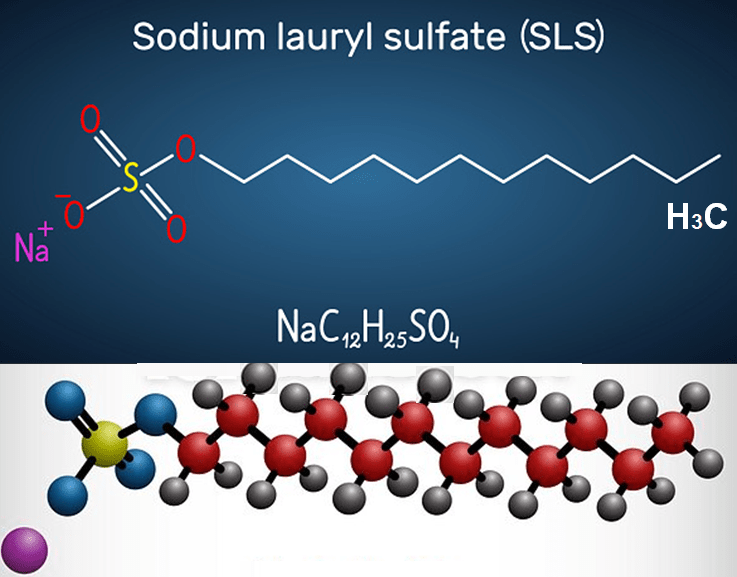
Sodium lauryl sulphate and its family of surfactants work so well and are so cheap that they’ve found their way into pretty much every type of cleaning product. They aren’t just in shampoo, but also washing-up liquids, laundry detergents and even toothpastes – that’s why your mouth fills with foam when you brush your teeth. Again the role of the foam is purely for show – Look, I’m washing my teeth! It says. The success of sodium lauryl sulphate ultimately led to it supplanting bars of soap as the main way to wash the rest of our bodies in the shower, not just our hair – it was the advert of so-called “body washes”. They were distributed in small bottles and squeezy containers like shampoos. And because the sodium lauryl sulophate family of surfactants are transparent, they look great in transparent bottles, especially if you color them and scent them, as you might shampoo.
The appeal of body washes wasn’t just aesthetic, though. When you’re in the shower or the bath, bars of soap have a disadvantage in that, as soon as they’re wet, they become incredibly slippery. If you’re taking a bath in an area with hard water, and you’re using a bar of soap, not only do you end up sitting in scum water as the soap with the calcium in the water, you also risk losing the soap altogether if it slips from your hand into the cloudy water. And if you’re in the shower when the soap slips out of your hand, it usually shoots off, pinging its way around the tub like a ricocheting bullet, potentially landing underfoot where you might step on it, lose your balance, slip and brain yourself. Not so with the shower gel.
Shower gel also has the advantage of being contained within a bottle. Bar soap has to sit somewhere, usually on an exposed surface, where it sloughs off its outer skin of foam and slimy scrum, giving it an unappealing, and certainly not very telegenic, appearance – unlike liquid soaps, which maintain their media-friendly appeal through every use. And even once it’s dry again, bar soap never quite returns to its solid dependable appearance; after just one use, the bar is misshapen.

In the 1980s a company called Minnetonka started thinking about ways of bringing liquid soap out of the bathroom and into toilets and kitchens. But they knew it needed to feel different – it couldn’t be like shampoo or body wash, and certainly not washing-up liquid – even though it would, in fact, be a very similar product. They had to sell it to people as new, and entirely appealing. They hit on the idea of pump dispensers, and that turned out to be a stroke of genius. Anyone who had previously worried about picking up a bar of wet soap that had already been used by the previous person in the toilet could now enjoy the seemingly pristine experience of having detergent dispensed straight into their palm. It didn’t catch on right away, though. Not everyone was impressed – for some it seemed to be an overly complicated solution to a non-problem.
But if the public were ambivalent about liquid soaps in the 1980s, the 1990s brought along something that tipped the balance firmly in their favour: a bacterium called Staphylococcus aureus that typically infects wounds after surgery and has, over time, developed strains that are resistant to antibiotics, making it very hard to treat. These strains were first discovered in 1960s, but by the 1990s Staphylococcus aureus that were resistant to treatment by the antibiotic methicillin had become an epidemic in hospitals. In the UK, methicillin-resistant Staphylococcus aureus (MRSA) infections accounted for 50% of all hospital infections. There were similarly high rates across Europe and the United States, leading to a sharp increase in hospital mortality. By 2006, the UK had seen 2000 deaths due to MRSA and hospitals were struggling to deal with spread of the bacterium. Fortunately, thanks to stricter handwashing regimes – in particular, requiring nurses and doctors to wash their hands after contact with patients – the rate of deaths has gone down in the past decade.
Outside the hospital, though, a public-health campaign began extolling the benefits of clean hands, and it hinged on the promotion of antibacterial soaps, which along with sodium lauryl sulphate and its cousin molecules, contain agents like triclosan, and antimicrobial molecule. These soaps were marketed as being better than traditional soap at preventing the spread of germs. The marketing was successful – the demand for antibacterial soaps was enormous, despite the fact that there was never any evidence that they were more efficacious than conventional soap and water. In fact, Dr. Janet Woodcock, director of the US Food and Drug Administration’s Center for Drug Evaluation and Research, said that certain antimicrobial soaps may not actually serve any health benefits at all. Consumers may think antibacterial washes are more effective at preventing the spread of germs, but we have no scientific evidence that they are any better than plain soap and water. In fact, some data suggest that antibacterial ingredients may do more harm than good over long term.
In 2016, antibacterial soaps were banned in the USA. But since then liquid soaps have infiltrated everywhere. Stripped of their antibacterial agents, liquid soaps now account for the majority of soaps bought in Europe and the USA. They’re still in our hospitals, our homes and yes, in aeroplane toilets as well. For instance the liquid soap you use in the toilets in aeroplanes contain sodium laureth suphate which is most likely made from palm kernel oil or coconut oil. These trees flourish in tropical climates and have become incredibly important to the global economy because they are easy to grow and have a high yield of oil, making them stable and profitable crops for any countries with suitable climates. 50 million tonnes of palm oil are produced annually and that goes into everything from cakes to cosmetics – next time you’re in a supermarket, take a look at the ingredients of biscuits, cakes, chocolate, cereal and on and on. You will very likely find palm oil in all of them.
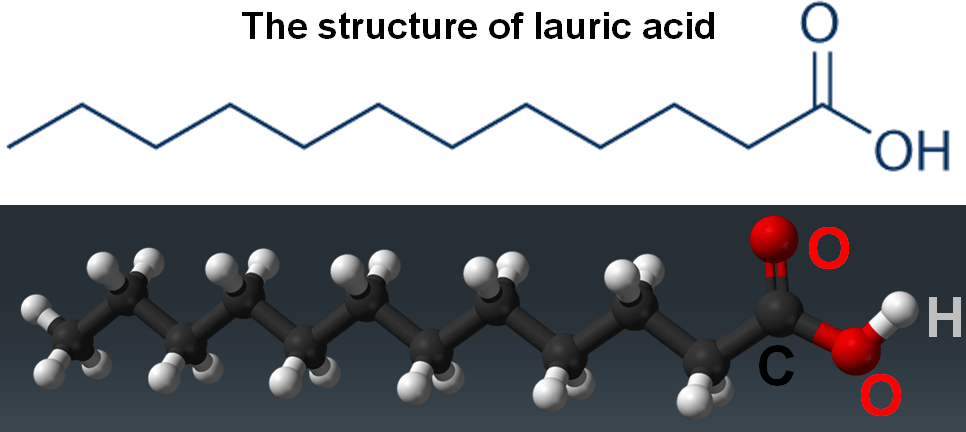
Palm kernel oil is particularly useful for making liquid soap because of its chemical composition; it contains a lot of lauric acid, a 12-carbon-chain molecule with a carboxylic acid group at the end. It looks a lot like a surfactant, but without a charged end. That’s easily fixed, though chemically speaking, its size is what is important; lauric acid, when used to make a surfactant, creates a chain molecule that’s much smaller than the ones found in normal soap, which are typically 18-carbon atoms long.
Lauric acid, being smaller, it is lighter and more effective as a foaming agent. In fact, it’s almost too good. Our delight with these liquid soaps has led to a huge increase in their production, and in demand for palm oil and coconut oil. This has, in turn, resulted in cutting down large sections of the rainforest in the countries where the oils are made, like Malaysia and Indonesia, replacing that immense biodiversity with a monoculture of palm trees. This has all sort of negative impacts, not least among them the destruction of habitat for wild animals, many of them already endangered, and the displacement of indigenous communities, who have been marginalized for centuries. Such is the demand for liquid soaps, though, and for the other uses of palm oil, that this process continues.
And to add insult to injury, detergents made with sodium laureth suphate, which we go to such lengths to make, can actually work too well for some people. They remove fats and oils so well that they cause skin irritation, like eczema and dermatitis. To prevent this, liquid soap manufacturers add modifiers and moisturizers to their soaps, which replace the natural oils that the sodium laureth sulphate pulls out of your skin. You could almost be glad, then that most of the liquid soap that’s used just ends up going down the sink without even interacting with your hands. Liquid soap manufacturers have tried to address this by increasing the viscosity of the soap, and also by creating dispensers that squirt the soap out not as a liquid, but as a pre-formulated foam, which is more useful. The foam dispensers are actually rather good, not just because they dispense the tiny amount of surfactant you need, along with a lot of air, but also because they’re finally created an active use for foam. It’s not just aesthetic, as it is in shampoos, body washes, toothpastes. In foam dispensers, the foam is the medium that carries the surfactant to your hands.
All in all, liquid soaps, of varying sorts, have become a 100-billion-dollar industry. We rely on detergents to keep ourselves clean and scented, to keep out clothes clean and scented, to keep our hair clean and scented; to wash our dishes, and perhaps most importantly in a highly populated world, as one of the most powerful ways of keeping ourselves healthy and stopping the spread of disease. But when we buy them we’re mostly paying for their marketing; the essential ingredients of detergents, the ones that do the cleaning; are cheap – all the more reason to consider how these products are being made, and their impact on forests in the tropics. Me, I love a bar of soap. It’s hand-sized and washing with it gives me the feeling of material contact, which I find reassuring and comforting.
Yes, bars of soap are hard to market, but that’s part of what I like about them – you buy a bar of soap because you need it, not because you think it’s going to make you a different, more successful, more desirable or sexy person.

Leave a comment