I hate dirt and the sticky things that remain on my hands whenever I work on something. Even if I eat a cake or I taste different sweets, after I eat and after i use the toilet, I cannot continue my day if I don’t wash my hands. The same feeling I have about dirty clothes, I just hate to feel the scum and dirt around me. I am sure I am not the only one, therefore in this article I wish to point some aspects about the importance of cleanliness more in details. To fight and remove dirt we need SOAP.
Soap is a miraculous substance. You can wash yourself with the clearest, purest, hottest water, but you will not get rid of any of the oily grimy muck that´s caked on to your skin. For most of history, we weren´t unduly worried about this. People smelt; they were dirty. No one cared. We had bigger problems, and no sense of why soap might be important. Which isn´t to say soap didn´t exist. Recipes for making soap on ancient Mesopotamian clay tablets have been found as far back as 2200BC, but the material has almost certainly been around for longer than that. The process described is similar to how we make soap today:
- take ashes from a wood fire,
- dissolve them in water, and
- boil the solution with melted tallow (animal fat) – and magically you have a basic soap.
While the Mesopotamians didn´t necessarily use soap to bathe, they did use it to clean wool before weaving it into fabrics. The soap removed the lanolin, a kind of grease, from the wool fibres. But why would you use fat to remove grease? The secret is in the ash water, the Arabic word for which is alkali, which literally means “from the ashes”. Alkalis are the opposite of acids, but both are highly reactive and can transform other molecules. In this case the alkali transforms the fats. One of the main constituents of tallow, is a molecule called a triglyceride. This has 3 tails that can be snipped off using alkali.
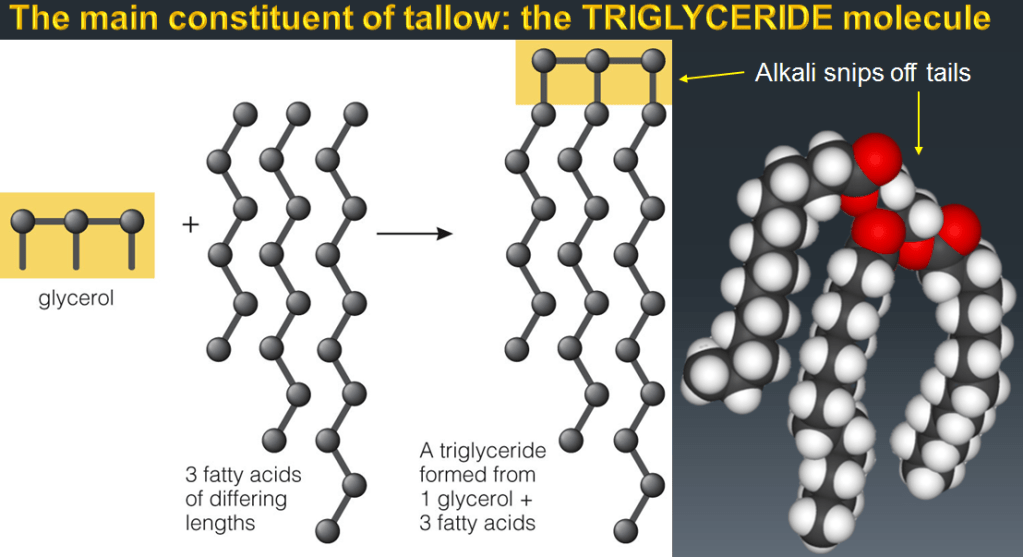
Fats, such as animal tallow, are made up of carbon molecules, with a 3-armed chemical structure of glycerides bound together at one end by oxygen atoms. The structure is completely different to water, which is much smaller H2O molecules. Water molecules are not just smaller than triglycerides, they are also polar, meaning the electric charges on the molecule are not distributed equally: there is a positive part and a negative part. This polarity is what makes water such a good solvent: it is electrically attracted to, and surrounds, other charged atoms and molecules, thus absorbing them. Water dissolves salt this way, it dissolves sugar this way, it dissolves alcohol this way. But fat and oil molecules aren´t polarized, so they can´t dissolve in water. This is why oil and water don´t mix.
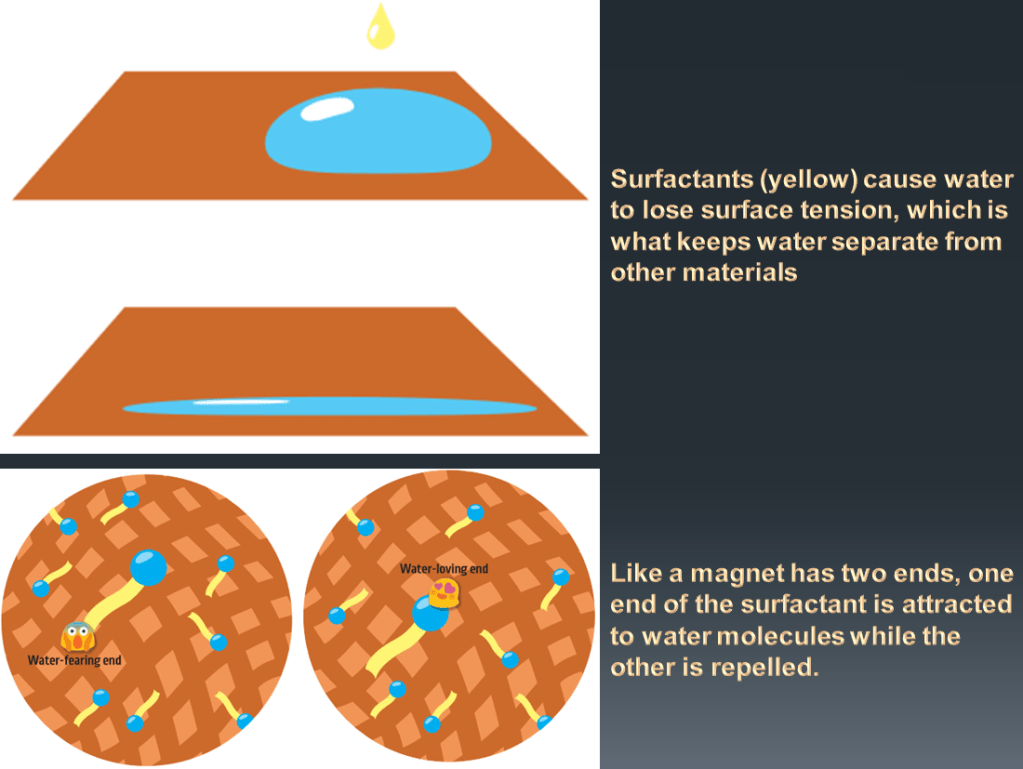
Have you ever seen a bead of water sitting on a surface? I am sure you have. You see it all the time on the wildows in your house or car after the rain stops. This is because water has a property called surface tension. This tension causes water to form a bead on the surface of things like glass or fabric. You can also see surface tension at work by placing a drop of water onto a counter top. The drop will hold its shape and will not spread. In order to clean the dirt on our clothes, the water needs to be able to reach the surface. Water is able to get to the surface if surface tension is reduced. To do this, we use a group of chemicals called surface active agents, or surfactants.
What is a surfactant?
Surfactants change how water behaves. When a surfactant is added, the surface tension is reduced. Now water can spread out and wet the surface (e.g., clothes, dishes, counter tops) we are trying to clean. Now let’s look at what happens on the surface.
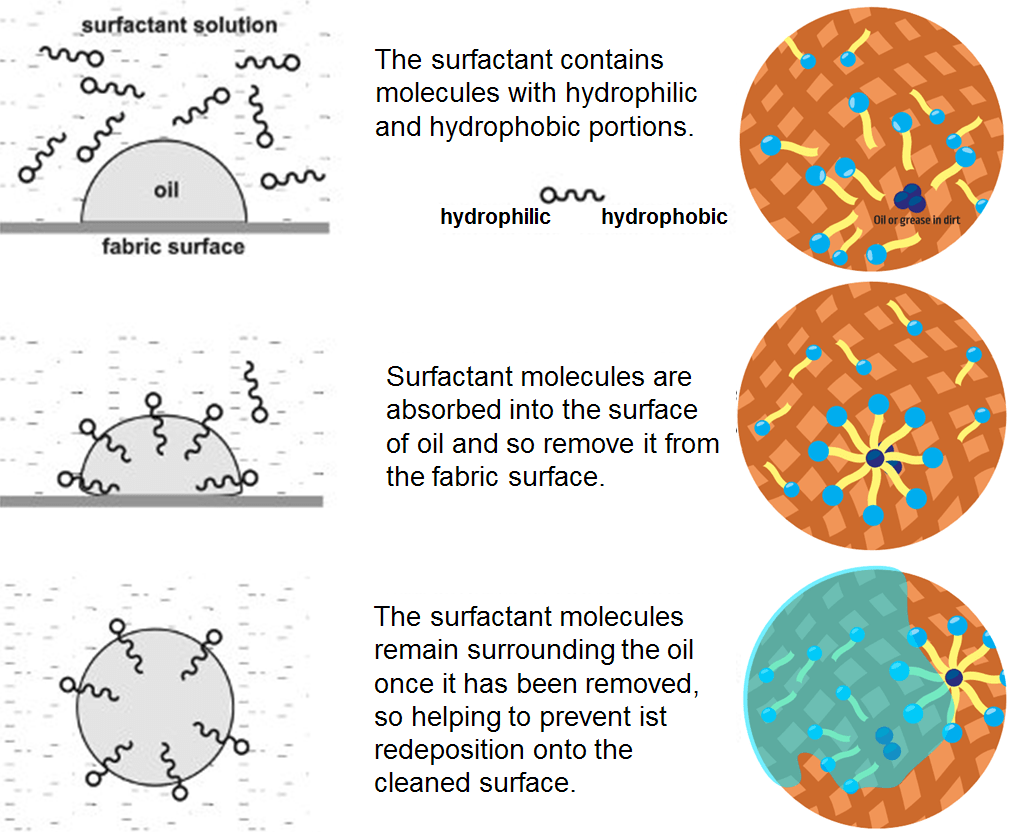
The alkali produced from wood ash splits into positive and negative components, though, so it dissolves in water. The resulting solution chemically reacts with the fat molecules, snipping off the 3 tails of the triglycerides, making them charged. This produces 3 soap molecules (called stearates). Importantly, these are hybrid molecules that have an electrically charged head, which likes to dissolve in water, and a carbon tail, which likes to dissolve in oils – it’s this hybrid nature that makes soaps so useful. The active ingredient in soap stearate, is showing its charged head, which is “water-loving”, and a carbon tail, which is “fat-loving”.

When soap molecules come into contact with a blob of oil, the carbon tail of the molecules immediately buries itself in it, thanks to their chemical similarities. But the charged head of the soap wants to get as far away from the oil as possible, so it ends up just sticking out of the blob. As more soap molecules do the same, they form a molecular structure that looks like a dandelion seed: a blob of oil surrounded by a cloud of soap molecules, with their electrically charged heads sticking out. Because the blob of oil or fat has now got a charged surface, it’s become polar, and so will dissolve happily in water. This is how soap cleans – it breaks up fat and oil residue on your hands and clothes into tiny spherical blobs, which can dissolve in water and be washed away.
How Surfactants Clean?
Once the surfactant is added to water, the water-fearing ends try to stay away from the water. They do this by organizing into the shape of a sphere with the water-loving ends on the outside and the water-fearing ends protected on the inside. This spherical shape of surfactants is called a micelle.
The micelle is important because it is what traps the soil. Remember, the inside of the micelle is hydrophobic and does not want to be near water. The soil is also hydrophobic, so it likes the environment the micelle creates. The attraction of the soil to the inside of the surfactant micelle helps loosen the soil from its surface. Once the soil lifts off the surface, it becomes suspended in the water in the micelle. This suspension is also known as emulsification of one liquid into another. Happy inside the micelle, the soil will not settle back onto the surface. Now that the soil is trapped in the micelle and the micelle is suspended in water, it is easy to wash the soil way. Remember the outside of our micelle loves water. So, as we rinse, the micelle floats away and we are left with a clean surface!
Soap cleans by the action of surfactant molecules, such as stearates. The fat-loving tail of the molecule is absorbed into oil, leaving the water-loving head sticking out. The cloud of water-loving heads surrounding the oil allows it to be dissolved in the water and so cleans a surface. The clean, dry feeling you get from washing your hands with soap comes from the soap removing oils from your skin. In contrast, soap is slippery precisely because of its own fatty nature – it’s basically modified fat. That’s why it slips out of your hand so easily. It is why soaps are used as lubricants, why, if you are trying to remove a ring off a swollen finger soap can slip it right off. Using soap to clean creates a special type of liquid; it’s dirty water, yes, but it’s made up not just dirt, but also of balls of fat. In effect, it’s one liquid suspended in another – an emulsion.
Emulsions
These suspended liquids are very useful because they allow you to suspend many different types of liquid in water.
Mayonnaise, for instance, is a very concentrated suspension of oil in water, where the ratio of oil to water is roughly 3:1. You make it by shaking the two together vigorously, until they form a cream. If you just did that, though, the liquids would separate, because, as we know, oils and water don’t mix. But if you add a soap-like molecule, it will stabilize the droplets of oil. For mayonnaise, the binding molecule comes from eggs. Egg yolks contain a substance called lecithin, which has a structure very similar to soap (with a fat-loving tail and a water-loving head), and when you add them to your oil/water mixture, they bind it all together and make mayonnaise. Egg yolk can also clean your hands, just as soap does, and there are plenty of shampoo recipes that use egg yolk as an essential cleansing ingredient.
Mustard is another substance that can emulsify oils – which is why if you add mustard to oil and vinegar, which otherwise don’t mix well, it will form a stable emulsion, also known as a vinaigrette. All these active substances work in the same way, and they all have a shared name: they’re interface molecules called surfactants.
But soap doesn’t just remove oil and fats; it also removes the bacteria that are attached to those oils and fats. Washing your hands with soap is the single most effective way to protect against bacterial infection and viruses. But despite the efficaciousness of soap as a cleaning agent, and its discovery so early in human development, the regular use of soap for cleanliness and personal hygiene is a modern phenomenon.
*************************************************************
Throughout history, different cultures took very different stances on the use of soap. The Romans didn’t really use it, preferring to scrape off their sweat and dirt mechanically, and then bathe in first hot then cold water, to get clean. Their public baths were an important part of their culture, and relied on a sophisticated engineering infrastructure to provide hot and cold water. In Europe, after the Roman Empire collapsed, the infrastructure that kept the public baths going fell into ruin, and so bathing went out of fashion. In crowded cities and towns, without access to clean water, bathing was increasingly considered a health risk. During the Middle Ages, many Europeans believed that diseases were spread through miasma and bad air. They thought that washing, especially with hot water opened up the pores and made a person more susceptible to diseases, such as the bubonic plague, also known as the Black Death. There was also a moral component associated with washing at this time, where to be holy like a hermit or saint involved rejection of comfort and luxury. Hence the more you smelt, the closer to God you might be perceived to be. These peculiar attitudes to cleanliness were not present in other parts of the world, and so visitors from the East would have found even the royal Europeans bewilderingly smelly and dirty, just as we would, looking at them from our modern vantage point. But the cultural norms of the past can often seem disgusting in hindsight. It wasn’t that long ago that smoking was quite normal, and the smell of smoke was pretty much everywhere, in offices, restaurants, bars and trains. There was at time when smoking was allowed even on aeroplanes. Now we look back with a mixture of horror and puzzlement as to how we got ourselves into that predicament. Seen in this light, the era of filthy, smelly Europeans is perhaps not so surprising.
As with smoking, the consequences of general uncleanliness were not just aesthetic. In the 19th century it was still normal practice for doctors to travel from bedside to bedside without changing their clothes or washing their hands after examining women during childbirth. This practice caused incredibly high rates of maternal and infant mortality during childbirth. In 1847 a Hungarian obstetrician, Ignaz Semmelweis, mandated that physicians scrub their hands with a chlorinated lime solution before touching patients, and saw the death rate fall from 20% to 1%. Despite this evidence, doctors were still reluctant to accept that they might be carrying infections on their hands, and transferring them to their patients, thus causing an enormous number of deaths. It wasn’t unti the 1850s when a British nurse, Florence Nightingale, took up her campaign for cleanliness that this attitude was really adopted, first in military hospitals, and then more widely. Crucially she gathered statistics and invented new types of mathematical charts to show her evidence to doctors and to the public, about the causes of disease and mortality. Gradually, as scientific evidence mounted, germ theory was accepted more generally by nurses and doctors, and hygienic washing with soaps are created equal, and soap’s new role in keeping people clean and healthy came at a time when industrialization and marketing were combining to create our modern Western consumerist culture. Soap was ready to transition from a commodity into a commercial product.
Laundry powder was one of the first industrial products based on soap. Everyone had to wash clothes, and the growing importance of hygiene and cleanliness shaped attitudes towards social status in the 19th century: if you wore dirty clothes to a party, or to church, or to any other religious gathering, you were deemed not just poor and low, but increasingly you were also held to be immoral. No longer was it true that being smelly and dirty was a signifier of virtue. Germs and disease were now associates with unclean habits. So in 1885, when the Reverend Henry Ward Beecher declared that ‘Cleanliness is next to Godliness’, he was voicing a widely held belief that morality and spirituality had aphysical manifestation, and that soap was an indispensable aid to this higher status. At the same time, the spread of both railways and newspapers was bringing people together, making it possible to spread one message across a nation – soap brands were able to become national institutions.

In the USA Procter &Gamble (P&G) became the most powerful presence in the soap industry. Founded in Cincinnati in 1837 by two English immigrants, William Procter and James Gamble, Procter & Gamble was in the business of selling candles and soap, both made using tallow from the local meat industry. But as the 19th century progressed, the candle industry declined – 1st because of the popularity of whale oil and then because of kerosene – while the market for soap grew. P&G invented Ivory soap and invested large sums in marketing it across the whole country, placing adverts in national newspapers and magazines. Then with the invention of the radio in the 1920s, P&G started sponsoring serial dramas, which were mostly listened to by women, alone at home during the day, washing clothes and doing household cleaning; the dramas were very popular and ended up being named after the product that sponsored them – “soap operas”.
The invention of the washing machine liberated people – primarily women – from the demanding social ritual of washing clothes, and with it came a whole new set of substances to clean our dirty laundry. Soap, which had been the primary way to wash clothes for almost 5000 years, suddenly got a chemical upgrade; it became detergent. Detergents are a cocktail of cleaning agents; they contain surfactants like soap, but also many other ingredients to make them more effective and less environmentally damaging. In soap, the charged, water-loving head of the molecule is attracted to the calcium in water, so if you live in a place that has hard water, the calcium will attach itself to the soap and form scum, just as it does on tea. Soap scum, though, looks slightly different – it’s the whitish substance you get on your hands when you wash them with a bar of soap. The scum isn’t just inconvenient; it also uses up the soap, so less of the bar is ultimately available for cleaning. It can also leave an unattractive grey residue on clothes.
What to do about soapy scum? – You have to make soap that’s less attracted to calcium. Chemists discovered a new set of molecules like soap, with a water-loving head and a fat-loving tail, but with these molecules they could carefully control the electric charges and make them less attractive to calcium: these were the new surfactants.
As demand for detergent grew, competition between manufactures became intense. Companies employed the best chemists they could find in the hope of creating better detergents. They developed detergents containing mild bleaches that could better preserve whites by reacting with the molecules responsible for brown stains, and snipping them up chemically. They also put fluorescent molecules into washing powder, called optical brighteners, that attach to the fibres of white clothes and stay there till after the wash. Optical brighteners absorb invisible ultraviolet light, and emit blue light, giving fabric that “whiter than white” look that so many detergent companies advertise. You can see how they work if you go to a nightclub: the ultraviolet lights over the dance floor activate the fluorescent molecules in your white clothes making them glow.
The range of surfactants expanded. Anionic surfactants (in which the water-loving head of the molecule is negatively charged as in soap) were created not just to avoid scrum formation and remove dirt, but also to keep dirt from redepositing itself on the clothes during the wash. Cationic surfactants (in which the water-loving head of the molecule is positively charged) were developed as fabric conditioners. And non-ionic surfactants (in which the water-loving head of the molecule is neutral) remove dirt even at low temperatures, and are less foamy than most other surfactants. Avoiding foam is important; foam does not help to remove stains, and it is inconvenient for washing machines to fill up with foam, as it is hard to get rid of. In fact, detergents often contain antifoaming agents, to suppress bubble formation. Biological enzymes are added to most detergents in an effort to reduce the environmental impact of washing clothes. The enzymes help to chemically snip up the proteins and starches you’d find in stains. They’re able to remove stains at lower temperatures, which makes low-temperatur washing machines much more effective, thus saving energy and money. We call the enzymes biological because they’re derived from natural enzymes found in living systems that do a similar job of degrading and mopping up unwanted stuff in the body. In Europe there are 2 kinds of laundry detergents: bio and non-bio. The bio-detergents contain enzymes, and while these clearly wash cleaner, the non-bio detergents are still available because of a persistent myth – that has never been verified – that bio-detergents cause skin irritation.

Leave a comment