The technology of CabonNanoTubes is still in early stage. We are not yet ready to fully benefit of this unique material at large scale. Some little progress has been made since professor Iijima created the first Cabon Nanotubes in 1991, but obviously we must do and learn more. It’s only when Carbon Nanotubes are grouped by purity, metallic or semiconducting, that they can be used effectively … which will ultimately advance all of those big promises that were made. Therefore Carbon nanotubes strength relies on creating a continuous perfect lattice of carbon atoms in a long tube, and that process is not something we have yet fully developed.
The Carbon Nanotubes as the one dimensional (1D) structures and carbon nanotubes is one of the famous allotropes of carbon actually.
The process of producing the microscopic structures of Carbon Nanotubes (CNTs) typically involves heating carbon-based gases to a point where mixed clusters of nanotubes form spontaneously as black soot. The production process itself is relatively easy yet the major problem standing in the way of the new technology, however, has been untangling metallic and semiconducting carbon nanotubes, since both are created simultaneously in the process of producing these microscopic structures. Only pure semiconducting or metallic carbon nanotubes are effective in device applications, but efficiently isolating them has proven to be indeed a challenging problem to overcome. Even when the nanotube soot is ground down, semiconducting and metallic nanotubes are knotted together within each grain of powder. Both components are valuable, but only when separated.
To understand this better, let’s first take a closer look at how Cabon Nanotubes are structrally made.
THE MOLECULAR STRUCTURE OF CARBON NANOTUBES.
Carbon nanotubes are tubes or let’s say cylinders made of carbon atoms with diameters typically measured in nanometers. Although not really made this way but we can imagine carbon nanotubes could be formed from graphene sheets. As we know, the graphene is 1 atom thick layer sheet of carbon atoms bonded in hexagonal Network. If we cut through that sheet we get graphene on ribbons and if we roll up that ribbon throughout its length basically what we are making is a carbon nanotube. Although carbon nanotubes are not made this way in practice, but that’s a nice picture to have in mind actually.
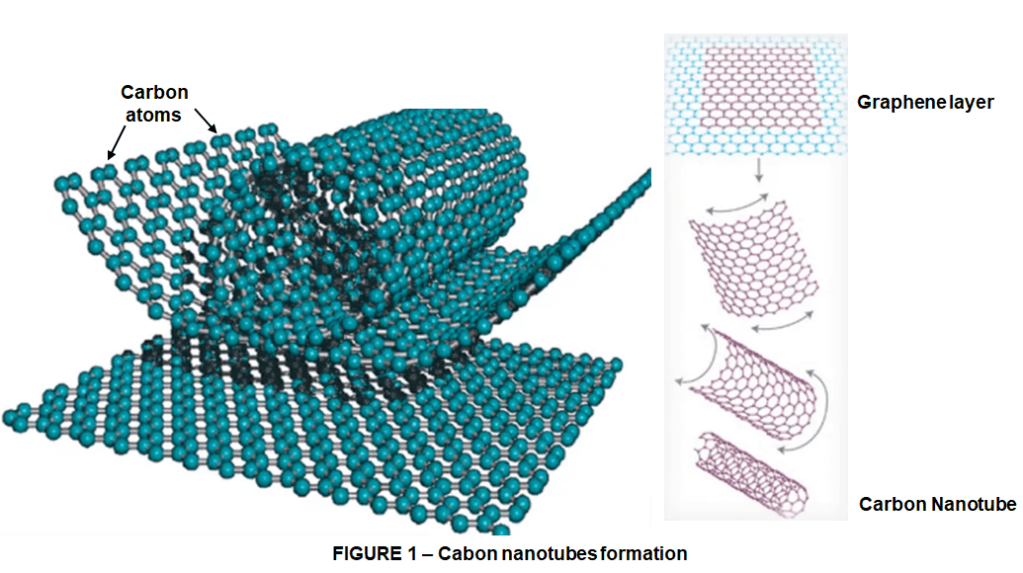
One of the first things that comes in mind about making carbon nanotubes from graphene is the cutting orientation because depending on the cut orientation different forms of carbon nanotubes are obtained. Let’s make it more clear as shown in figure 2.
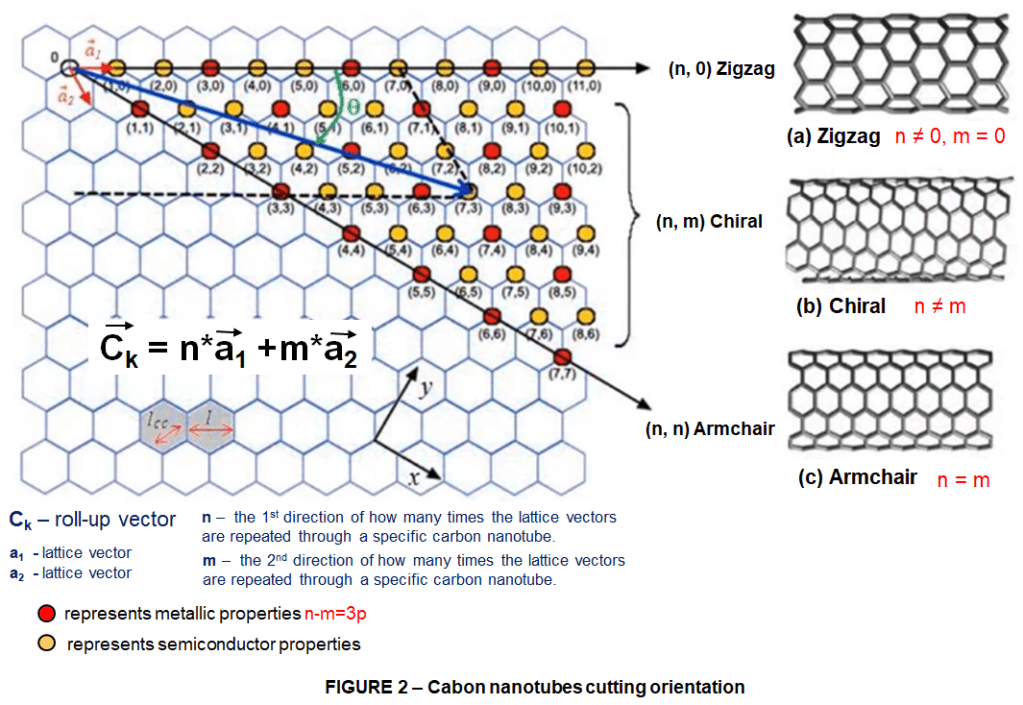
If we cut graphene in direction (a) and then roll it up from the other side our carbon nanotubes is going to have the exact edge and because of that this form of carbon nanotubes are called Zigzag carbon nanotubes.
If we cut graphene in direction (c) and roll it up through another vertical direction what is concluded is so-called Armchair carbon nanotubes.
And finally the third possibility to cut the graphene layer in direction (b) results in neither zig-zag nor armchair but a new form named Chiral Carbon Nanotubes.
These are basically the 3 main types of CNT based on the cutting orientation, furthermore there are a couple of options and also carbon nanotubes could be larger or smaller depending on how much wide we cut through the graphene.
To keep these carbon nanotubes recognizable; we also need to think about a naming method. We can do this through defining the roll-up vector of lattice as follow:
Ck = n*a1 +m*a1
Where :
- a1 and a2 = lattice vectors
- N and M = the directions of how many times is vectors are repeated through a specific carbon nanotube.
Now let’s see how this is working. Let’s look again at Figure 2.
The a1 vector as shown in the (a) direction is a vector representing moving one insert through this zigzag line of graphene sheet and n shows how many times this a1 vector is repeated by our card for that from the sheet.
The a2 vector is defined with respect to a1 representing oriental zigzag movement as show for directions (b) and (c) and m shows how many times this one is repeated.
So for example if we want to make a zigzag carbon nanotube we cut graphene by a1 vector and roll it up by the other side right in this case we have repeated vector a1,10 times and no repeat of vector a2.
Here the notation in parentheses is going to be like 10 – 0. Under this notation system zigzag carbon nanotubes have always m=0 and n should have some value.
Regarding Armchair carbon nanotubes n and m are always equal, like here we have more 7 times by a1 and 7 times by a2 and thereby we get 7-7 armchair carbon nanotube and when n ≠ m we are talking about a chiral carbon nanotube
In this way we can follow exactly how different structures of CNT occur.
Considerably in terms of conductivity not all of carbon nanotubes contain the same characteristics, some represent metallic behavior while some others are semiconductors.Why is that so? Well… That’s because each edge shape enforces different quantum confinement in carbon nanotubes, leading to different conducting behavior. In general for a given n and m nanotube, if n = m the nanotube is metallic if n – m = 3p and n ≠ m or neither of them are 0, then the nanotube is quasi metallic with a very small bandgap. otherwise the nanotube is a moderate semiconductor.
MANUFACTURING CARBON NANOTUBES.
Currenly the most promising method for industrial scale production of high purity carbon nanotubes is Chemical Vapor Deposition.
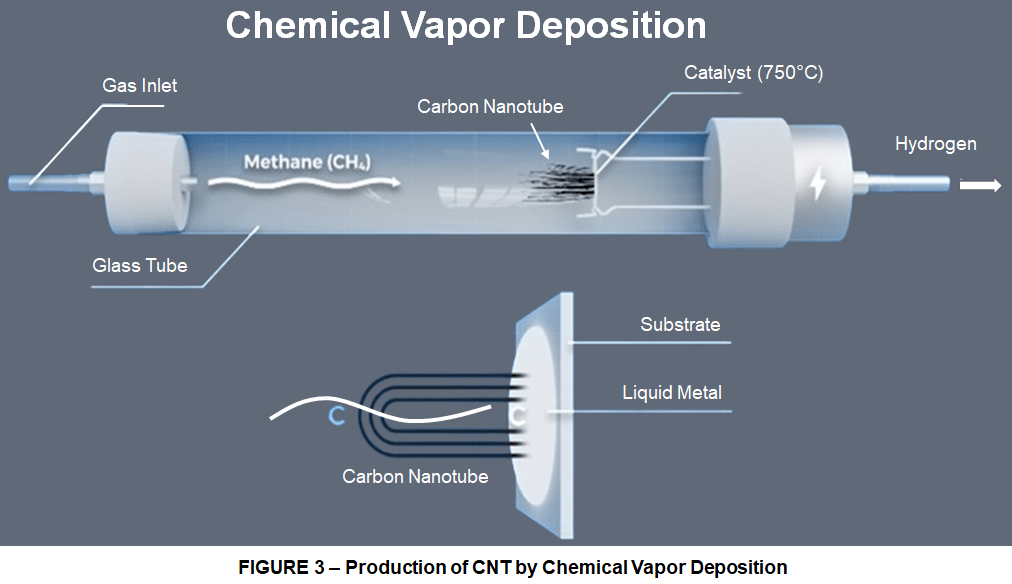
In this manufacturing method, a precursor gas containing carbon, like methane (CH4) is introduced into a vacuum chamber and heated. As the heat increases inside the chamber the bonds between the carbon (C) and hydrogen (H) atoms begin to decompose. The carbon then diffuses into a melted metal catalyst substrate. This then becomes a metal-carbon solution, which eventually becomes supersaturated with carbon. At this point the carbon starts to precipitate out and form carbon nanotubes, while the hydrogen bi-product is vented out of the chamber to avoid an explosion. The research here is focused on increasing the length of these nanotubes while not sacrificing their structure, yield or quantity. While some labs have gotten individual tubes as long as 50 cm it’s been a struggle to get larger bundles of tubes, which are called forests, to a length greater than 2cm. This is because the catalyst is guaranteed to deactivate at some point during the growth process, terminating the growth of the nanotube. The key to growing longer nanotubes is minimizing the probability of the catalyst deactivation
But there are good news. In 2020 reasearchers from Waseda University in Japan, led by professor Hisashi Sugime developed a method to grow a forest of super-long Vertically Aligned Carbon Nanotubes (VACNT) reching up to 14cm in length, 7 times longer than anyone else, using a new method of Cold-Gas Chemical Vapor Deposition that managed to keep the catalyst active for 26 hours.
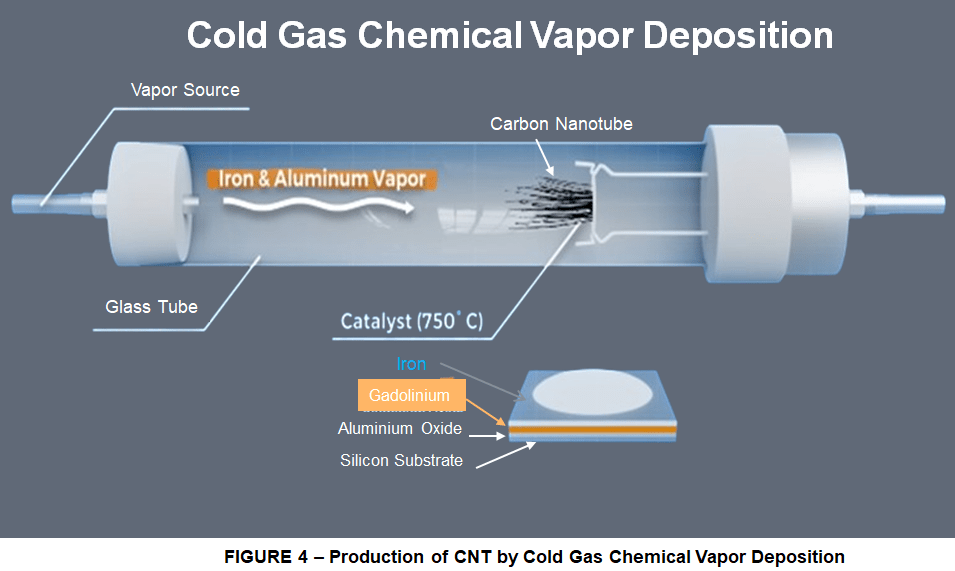
They did this by adding a gadolinium (Gd) layer to a conventional iron-aluminuim oxide (Fe/Al2Ox ) catalyst, coated onto a silicon (Si) substrate. This Gd layer prevented the deterioration of the catalyst to a certain extent, allowing the forest to grow up to around 5 cm in length Then using a lower chamber temperature, they heated it to 750°C and supplied it with small concentrations (parts-per-million) of iron (Fe) and aluminum (Al) vapor. These factors combined managed to keep the iron-aluminium oxide catalyst active for much longer. Various analyses to characterize the grown CNTs showed that they were of high purity and competitive strength.

This method is a major leap forward that could allow carbon nanotube products to begin entering the market, but we are still a long way from bigger projects such as for example the space elevator.
HOW TO ISOLATE CARBON NANOTUBES
Now once the CNT are produced, in order to use them efectivelly we must separate the metallic ones from the semiconductor versions. This is not an easy task at all. However; researchers around the world have spent years trying to find effective and efficient ways to isolate carbon nanotubes and unleash their value. Over the past few years there have been some breakthroughs on how to effectively isolate carbon nanotubes after manufacturing, which has helped to push Carbon Nanotubes back into mainstream technology.
One of the first idea was to create polymers that could allow semiconducting CNTs to be dissolved and washed away, leaving metallic nanotubes behind. Unfortunately there was no such process for doing the opposite, namelly: dispersing the metallic nanotubes and leaving behind the semiconducting structures.
Until 2016, when finally Researchers at McMaster University in Canada adapted a polymer process, which had been popularly used to reduce carbon nanotubes to only metallic components and they managed to find a way to reverse the process, leaving semiconducting nanotubes behind and intact while making it possible to disperse the metallic nanotubes. Now the next step, is to exploit this discovery by finding a way to develop even more efficient polymers and scale up the process for commercial production.
Another successfully research on this topic happened in 2018 when a team of researchers led by professor Jiaxing Huang from Northwestern University Chicago/ USA used the common chemical solvent, cresol (C7H8O), to isolate nanotubes. It is long known that aggregated tubes such as CNT are hard to disperse in solvents, and if you cannot get a good dispersion, then you won’t be able to make high-quality nanotube thin films that many application rely on. An initial approach used by previous researchers in order to bypass this problem was to use additives to coat the nanotubes; which chemically alter their surfaces and forced them to separate. Although these methods do work as well, they leave behind residues that alter the nanotubes surface structures, which in turn can blunt their desirable properties. By contrast using cresol, the team of professor Huang found that this solvent does not deteriorate carbon nanotubes’ surface functions. After separating the entangled tubes, researchers can simply remove the chemical by washing it off or heating it until it evaporates.
And that’s not all. After unlocking a new way to make carbon nanotubes in higher and higher concentrations, Huang and his team discovered new forms of the material as well. Namely as the concentration of carbon nanotubes increases, the material transitions from a dilute dispersion to a spreadable paste to a free-standing gel and finally to a kneadable dough like a kind of thick moldable gel.
These various forms can be molded, reshaped or used as conductive ink for 3D printing. The dough state of nanotubes is fascinating,It can be readily shaped and molded into arbitrary structures just like playdough. Essentially, this solvent system now makes nanotubes behave just like polymers. It’s really exciting to see cresol-based solvents make once hard-to process carbon nanotubes as usable as common plastics.
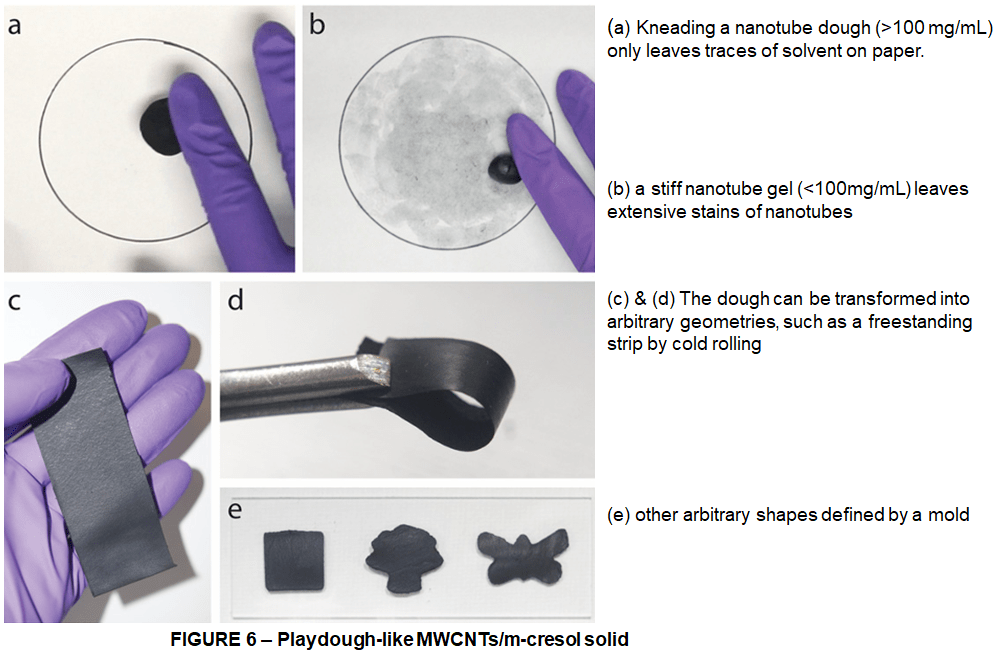
It’s these types of breakthroughs that are going to take us from bulk manufacturing, all the nanotube types tangled together, to isolated mass-scale manufacturing of Carbon Nanotubes. If one day we’re ever going to make that space elevator, we’re going to need a lot of isolated nanotubes.
Leave a comment