Carbon is a very familiar element. It’s in everything we eat, sleep on and step over. It is the element that holds our DNA together. It forms the carbohydrates, proteins and lipids that we depend on to build and fuel our bodies. It’s the basis of life as we know it. It’s ubiquity in our lives is a result of its versatility. It’s chemical properties allow it to take many different shapes, each impacting it’s material properties in diverse and unique ways. Different structural atomic arrangements of atoms of the same element are called allotropes. There are of course a lot of elements in Periodic Table which can create allotropes but by far the most versatile and studied are the carbon-based ones.
These materials occur naturally on Earth, they have unique properties that might have huge potential for many application, but for now except graphite and diamonds (more ore less), for the rest of carbon allotropes we don’t have yet the proper technology to use and produce them easily in the quantity and quality we would like to have. We must keep learning and one day when we’ll master the knowledge good enough things could certainly change.One such material that gained a lot of traction in the last 25 years is of course a carbon-based structure, namely the CARBON NANOTUBES.
HOW CARBON NANOTUBES (CNT) WERE DISCOVERED?
The history of CNTs is almost equivalent with the history of nanotechnology. If we really go back in time CNT have been found for example in pottery in from Keeladi village in India dating around 600 BC. though it is not known how they formed or whether the substance containing them was employed deliberately. There are numerous such examples since medieval times until 20th century linked to the existance of nanomaterials. Therefore the basic concepts of nanotechnology are not so new. However the real breakthrough started much later, namely by mid on 1950s. and became more obvious during 1970s. The same goes with CNT. Although CNTs were first observed in the late 1950s, formed by two layers of graphene, not exactly catchy names, but very accurate, the “holy grail” moment happened in 1991 when the Japanese physicist, Sumio Iijima, conducted a momentous experiment. An experiment that introduced the world to a material so strong that it could revolutionize how engineers approach design. It happened like this: Taking two graphite rods as electrodes, Sumio applied a current across the rods. A spark arched between them and with it a cloud of carbon gas puffed into existence, vaporizing the tip of the anode rod. As the carbon laden air settled on the chamber walls it formed a thin layer of black soot, within it a strange new material appeared, as tiny single layer straws of carbon. Hence here we have it, Sumio Iijima had just created carbon nanotubes.

That was a moment on a par with the discovery of microscopic bacteria; the world suddenly seemed a much more complex and extraordinary place than we had imagined. It wasn’t just living organisms that could self-assemble into complex structures, the non-living world could do it too. Since then an obsession with the production and examination of nanoscale molecules gripped the world, and nanotechnology became fashionable.
Laboratory testing of these mysterious little tubes in the following years would reveal that these nanometer-wide hexagonal lattices of carbon had the strongest tensile strength known to man, and this was just as one of the many incredible material properties they displayed. Carbon nanotubes are likewise light, conductive and biocompatible. It soon became clear that the carbon nanotube had the potential to be the building block of futuristic new technologies.
The most efficient computers, transformative medical devices, synthetic muscles, or perhaps the most ambitious of all, space elevators, the dream of countless sci-fi authors, Carbon nanotubes has promised to be the catalyst for the next revolution in technology. But, putting this revolutionary material to work will not be easy. It turns out that building a fibre, that is actually a single molecule, of any significant length is incredibly difficult. To understand this fascinating molecule, let’s dive into the chemical makeup of carbon nanotubes.
WHAT ARE CARBON NANOTUBES?
Carbon nanotubes are essentially molecular sheets of carbon atoms, also known as graphene, which are arranged in a hexagonal lattice or honeycomb arrangement – a bit like chicken wire. These sheets are organized in layers and manipulated into a cylindrical shape. So basically, graphene is the basis for Carbon Nanotubes. You can’t have Carbon Nanotubes without graphene. Now, these nanotubes can be designed and arranged in many ways, structurally they can be:
* Single-Walled Carbon Nanotubes (SWNTs) are made of, as you might imagine, a single layer of graphene. The way in which they are rolled can impact their conductivity.
* Double-Walled Carbon Nanotubes (DWNTs) are a combination of Single-Walled and Multi-Walled – with thermal, chemical and conductivity properties that are intermediate compared to the other two.
* Multi-Walled Carbon Nanotubes (MWNTs) are an extension of Single-Walled. They’re made-up of multiple layers of graphene which better insulates their thermal and chemical properties compared with Single-Walled tubes. Multi-walled carbon nanotubes consists of several interlinked nanotubes with diameter reaching more than 100 nanometers. Their lengths can reach several micrometers or even millimeters.

Depending on how the hexagonal lattice is rolled, it affects what’s called the chiral angle of the final tube, that’s how that hexagonal pattern spirals along the tube. If we follow the pattern across the diameter of the tube, we’ll see three basic types:
- armchair
- zig-zag
- chiral nanotubes.
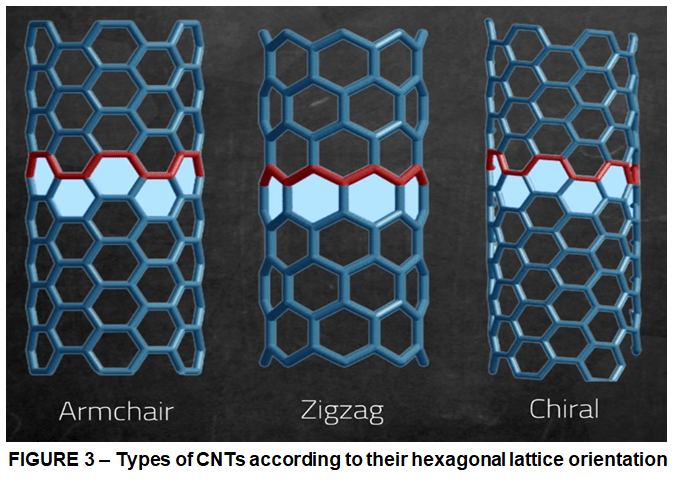
The orientation can make it act as either a metallic (therefore conducting) or semiconducting material. For instance compared to single walled carbon nanotubes. Pure multi-walled carbon nanotubes are always metallic. However despite the complexity of their molecular architecture, all these carbon nanotubes had a peculiar property: they could self-assemble. They needed no outside help in order to form these complex shapes, nor did they need high-tech equipment. They could do it in the smoke of a candle. This actually what happened also in the experiment described above. Therefore when manufacturing nanotubes, we usually end up with a mixture of types. The nanotubes have a tendency to clump into an entangled mess. And separating them apart has been the big challenge for mass-scale manufacturing. Remember, they’re only a few nanometers in diameter, or about 10,000 times smaller than the width of a human hair.
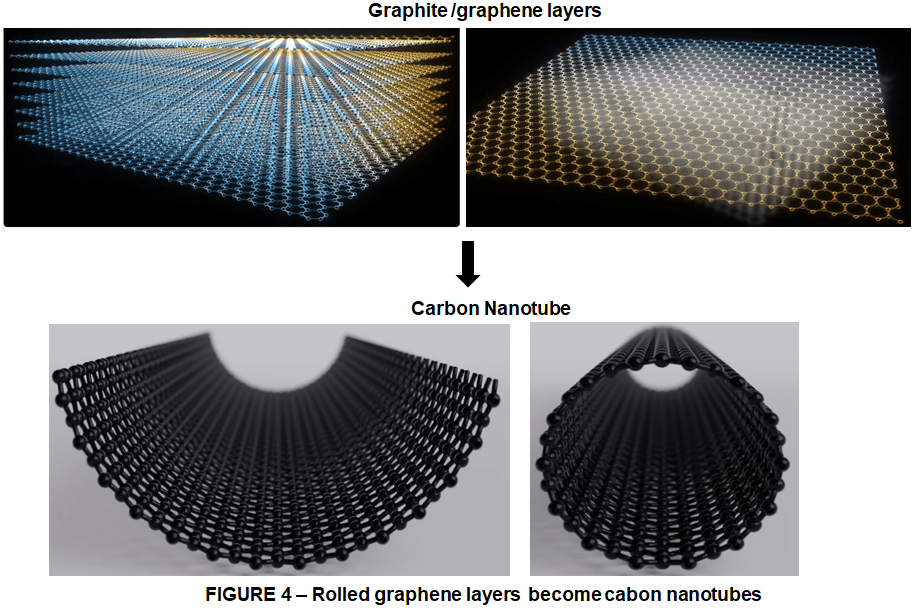
Now what is interesting is that carbon nanotubes take the same repeating hexagonal structure as graphite. The ends of the sheets are simply loops and connect with themselves to form a tube, and this structure is what gives carbon nanotubes their incredible strength. Researchers found that single-walled nanotubes have strength similar to that of graphite, about 130 GPa (Gigapascals). For the non-engineers in the crowd, let me rephrase that. It’s a lot. Namely it’s about 100 times greater than steel, and to boot it’s vastly lighter. If this material could be feasibly manufactured into a single extremely long fibre, it could potentially open up entirely new design possibilities. Just for a quick perspective overview we could literally manufacture everything from composite materials for large metallic parts like bikes, boats, and cars, to highly efficient transistors, nano-inks, bio tech, and even space elevators.
However, after years of expected market roll-outs, carbon nanotubes still haven’t made the splash in the tech world that they had promised. As usual, the hype set expectations too high … almost as high as the space elevator … so excitement dampened as time went on. Why exactly? Well…as awesome this looks the Carbon nanotubes are notoriously tricky to process — especially in large quantities. Being about 10,000 times thinner than a human hair, the wiry, tube-shaped structures are said to be stronger than steel and conduct heat and electricity far better than copper. But when mass produced — usually in the form of powders — the tubes twist and clump together. This complication is a major barrier to the material’s widespread applications.
Likewise in terms of fabrication scientists have now developed methods to control the synthesis of carbon nanotubes to obtain regular structures with specific properties. But this too, it is still is a not an easy job to do. CNTs can be current made at the order of few millimeters at the maximum length and often times the CNTs are not totally pure, In fact there are some serious issues in terms of widely using carbon nanotubes in practice. There are challenges like precise fabrication, mass production, integration at high volume besides the concerns that has been associated with toxicity, There are also issues in terms of the production cost which is currently extremely high, therefore not really profitable to allow large scale production.
Hence the key problem why CNT are still not yet largely used in application is not necessarily the production of Carbon Nanotubes themselves – that has been available long before – instead is the precision and separation of the different types of CNT (metallic and semiconducting) during manufacturing. And of course the production cost must notably decrease as well.

Leave a comment