Gas is the state of matter with no fixed shape and volume. That means the particle in a gas aren’t particularly attached to one another being in a constant state of motion either inside of their container or as dispersed in space. However the behavior of matter in gaseous state is strongly influenced by at least 4 main characteristics. These are:
- The amount of gas, namely the quantity of molecules involved (n);
- The Pressure (P) as the amount of force the gas exerts on its containter;
- The Volume (V) as the amount of space the gas occupies;
- The gas Temperature (T).
Each of these 4 characteristics are established by a law, and therefore we have the 4 laws of ideal gas. Next I’m going to present how these laws operate.
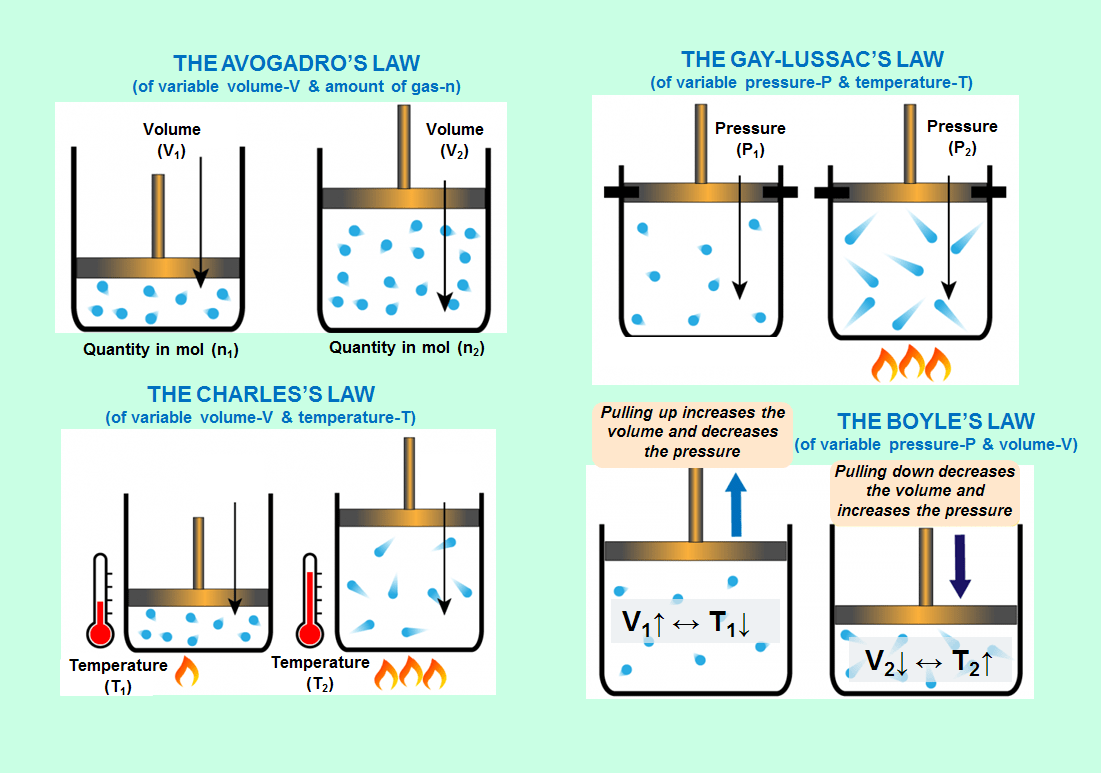
THE AVOGADRO’S LAW (of variable volume & amount of gas) (V & n)
In 1811, Italian scientist Amedeo Avogadro stated that:
“at the same temperature and pressure, equal volumes of all gases contain the same number of molecules, regardless of their chemical nature and physical properties”.
That means if the amount of gas increases, then so does its volume. For example, even though molecules of chlorine (Cl2) gas have a mass about twice that of oxygen, there will be the same number of molecules of each in containers of the same size and at the same temperature and pressure.(as shown in figure 1)
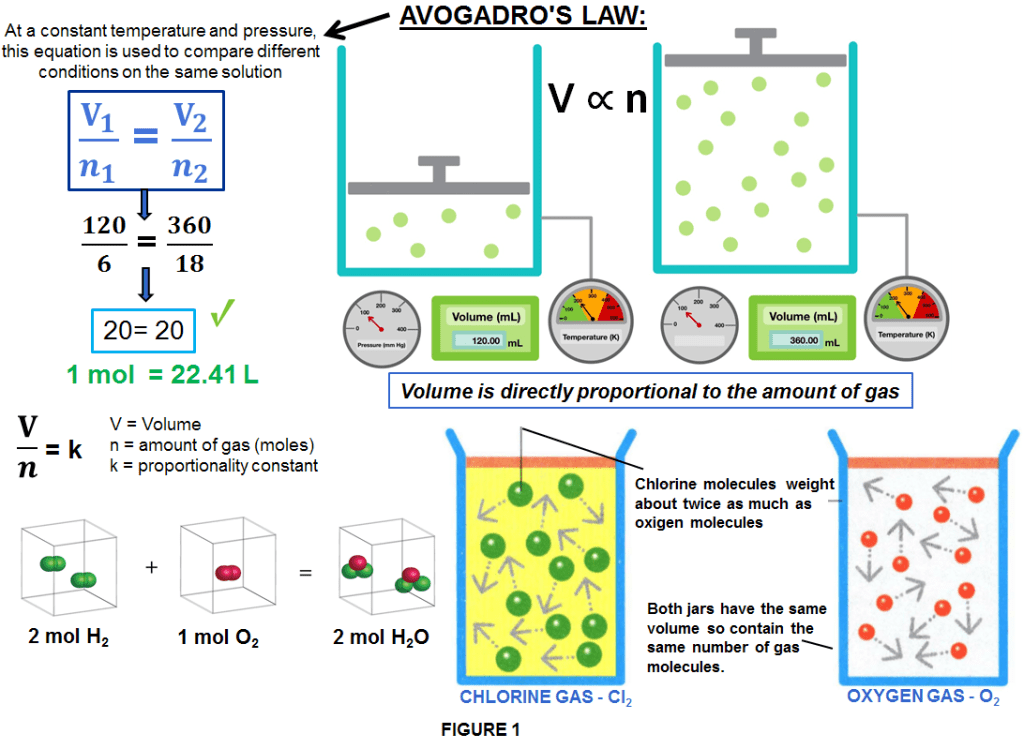
Therefore the Avogadro constant, was established as commonly denoted as NA and is an SI defining constant with an exact value of:
NA = 6.02214076×1023 mol-1 (unit of reciprocal moles).
The NA is the number of molecules of any gas present in a volume of 22.41 L and is the same for the lightest gas (hydrogen)(H2) as for a heavy gas such as carbon dioxide (CO2) or bromine (Br2).
THE CHARLES’S LAW (of variable volume & temperature)(V & T)
In 1802 the french chemist Joseph Louis Gay-Lussac referenced data gathered by his countryman, Jacque Charles, that:
“the volume and temperature of a gas have a direct relationship: As temperature increases, volume increases when pressure is held constant”
This means, if the volume of a gas is not restricted (by a rigid container, metalic for example), a gas will expand as it is heated and the gas molecules gain more energy. The higher the temperature of the gas, the greater its volume.(as shown in Figure 2).

For example, if the air in an inflated dinghy is heated by the Sun , the air will expand and inflate the dinghy more. This means that heating a gas increases the kinetic energy of the particles, causing the gas to expand. In order to keep the pressure constant, the volume of the container must be increased when a gas is heated.This law explains why it is an important safety rule that you should never heat a closed container. Increasing temperature without increasing the volume available to accommodate the expanding gas, means that pressure builds up inside the container and may cause it to explode. The law also explains why a turkey thermometer pops out when the turkey is done: The volume of air trapped under the plunger increases as the temperature inside the turkey climbs.
THE GAY-LUSSAC’S LAW (of variable pressure & temperature)(P & T)
Joseph Louis Gay-Lussac was a French chemist and physicist who discovered in 1802 that:
“assuming the volume and amount of moles remains constant, a gas’s pressure is directly proportional to temperature.That is,if you keep the volume of a gas constant (such as in a closed container), and you apply heat, the pressure of the gas will increase”
This means, if the volume of a gas remains constant in a rigid container – such as a diver’s air tank, for example – increasing the temperature of a gas increases its pressure. (as shown in figure 3)

This is because the gas molecules have more kinetic energy, causing them to move faster at higher temperatures, hitting the inside of the container more often and with more energy, so raising the pressure.
Another very common example is the air in car tires. When you drive a car, the air pressure on the inside of car tires changes. After driving, the air pressure in a car’s tires goes up. This is because friction (a contact force) between the tires and road causes the air inside the tires to heat up. The air cannot expand because the tires are essentially a fixed-volume container, so the pressure increases – this is Gay-Lussac’s Law!
THE BOYLE’S LAW (of variable pressure & volume)(P & V)
Robert Boyle, a famous English chemist, discovered in 1662 that:
“if the temperature and amount of a gas remains constant,volume and pressure have an inverse relasionship; that is increasing the pressure on the gas reduces its volume. Conversely, reducing the pressure on a gas increases its volume”
For example, if you doubled the pressure on a gas (increase the pressure two times), its volume would decrease by half (decrease the volume two times). The opposite is also true. If you reduced the pressure on a gas by 3.5 times, then its volume would increase by 3.5 times. This law is an example of an inverse relationship – if one factor increases, the other factor decreases.That is why bubbles expand as they rise to the surface of a liquid. This is know as Boyle’s law. As shown in Fig 4.

Specifically this means that by increasing the amount of space available will allow the gas particles to spread farther apart, but this reduces the number of particles available to collide with the container, so pressure decreases. In reverse, by decreasing the volume of the container forces the particles to collide more often, so the pressure is increased.
Another good example of this is when you fill a tire with air. As more air goes in, the gas molecules get packed together, reducing their volume. As long as the temperature stays the same, the pressure increases.
All these 4 gas laws assume that all gases behave like an “ideal” gas. In such a gas, there are no interactions between individual gas particles, the particles move randomly, and they take up no space. Even though no real gas has these features, the gas laws do show how most gases behave at normal temperatures and pressures. Because temperature and pressure vary from place to place, scientists use a standard reference point, called Standard Temperature and Pressure (STP), in calculations and equations.
- Standard temperature is T = 0°C – the freezing point of water and
- Standard pressure is P = 1 atmosphere (atm) — the pressure exerted by the atmosphere on Earth at sea level.
Leave a comment